 
The pH and the crowding agents had the greatest influence on the reaction enthalpy change. The reactions were monitored calorimetrically and the equilibrium concentrations were evaluated using the equation of state ePC-SAFT. 
Crowding by macromolecules was simulated using polyethylene glycol (PEG) and bovine serum albumin (BSA). 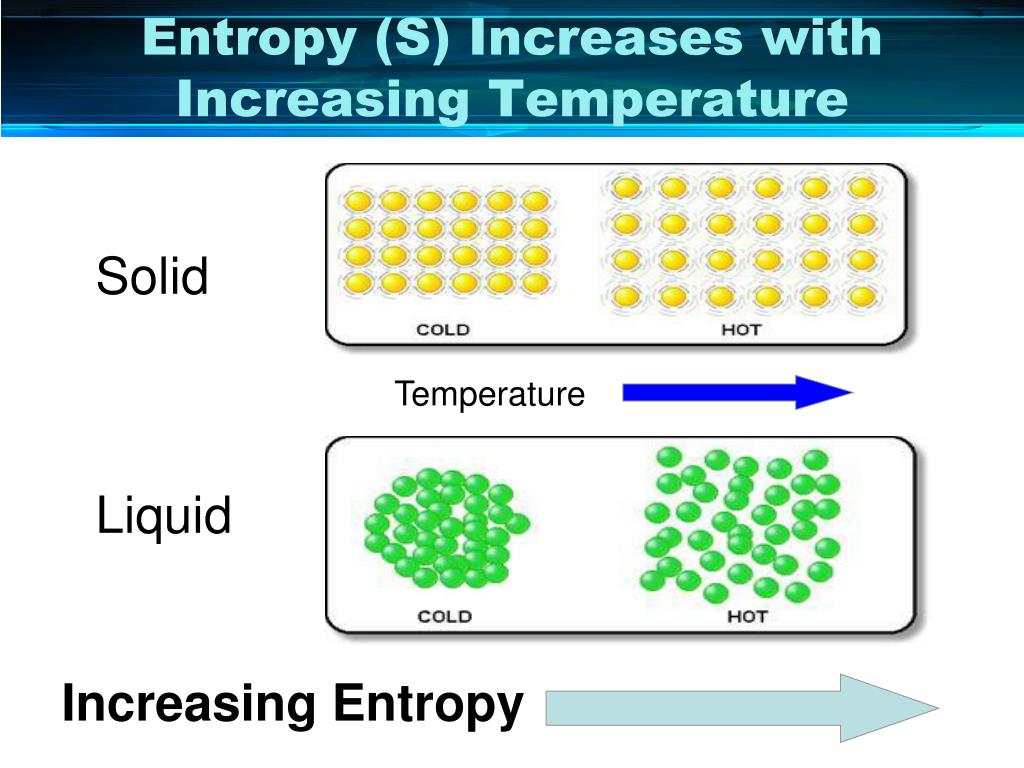
While in part I kinetic models based on irreversible thermodynamics were tested, here in part II, the influence of the presumably most important cytosolic factors was investigated using two glycolytic reactions (i.e., the phosphoglucose isomerase reaction (PGI) with a uni-uni-mechanism and the enolase reaction with an uni-bi-mechanism) as examples. This is because in this state, the system is unstable (the essence of a chemical reaction is to move from states of lower stability to that of higher stability).For systems biology, it is important to describe the kinetic and thermodynamic properties of enzyme-catalyzed reactions and reaction cascades quantitatively under conditions prevailing in the cytoplasm. To a disorderly state is always positive –įrom a disorderly state to an orderly state, entropy is always negative –Ĭhemical reactions take place more easily at a state of high disorder (i.e. Note: The entropy change involved when a system changes from an orderly state Change in Volume Occupied – when a gaseous substance decomposes into another gas of smaller volume, entropy decreases but entropy increases if the product is of higher volume. ∆S = +ve, but if two or more substances compose into a single substance, entropy decreases, i.e.ģ. Decomposition or Composition – when a substance decomposes into two or more substances, entropy increases, i.e. ∆S = +ve, but if the change is to a state of lower energy, entropy decreases, i.e.Ģ. Change of State – when a substance changes to a state of higher energy, the entropy increases, i.e. To Determine the Direction of Change in Entropyġ. When a gas is mixed with another without a change in total energy (i.e., without a reaction occurring), the molecules of the two gases which were initially separated diffuse into each other and the probability of a particular molecule of either gas occupying a given volume of space decreases during the mixing – this means that the system becomes more disordered - increase in entropy. salts) dissolve readily in water even though their dissolution in water results in a state of higher energy (since energy must be absorbed to bring about the dissolution). This is also why most crystalline solids (e.g. Therefore, to melt ice, heat is absorbed from the surroundings. A state of higher disorder (liquid) is also of higher energy than the state of lower disorder (ice). Possible to melt a lump of ice - since the molecules will assume a more disordered state in the liquid than in the solid state (ice). Thus, systems tend to be more disordered in their final state than in their initial state. I.e., gaseous systems show much greater disorder than liquid systems, which in turn show greater disorder than solid systems. It is the natural tendency for any system, when left to itself, to attain the highest possible state of disorder (randomness).ĭuring change of state, systems tend to a state of greatest disorder (high entropy) as the solid changes through the liquid to the gaseous state. The entropy of a system is a thermodynamic function, which measures the degree of randomness or disorder in the system. Free Practice Tests Entropy As Order-Disorder Phenomenon 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
